- Online only



Let us notify you about our product updates.
Venor®GeM qEP utilises quantitative, real-time PCR (qPCR) for fast (ca. 3 h) and reliable screening of cell culture supernatants for mycoplasma contaminations. The kit can be used in combination with any type of real-time PCR cycler able to detect the fluorescence dyes FAM™ and HEX™.
All Mollicutes (Mycoplasma, Acholeplasma, Spiroplasma) species so far described as contaminants of cell cultures and media components are specifically detected by amplifying a highly conserved rRNA operon, or more specifically, the 16S rRNA coding region in the mycoplasma genome. The mycoplasma-specific amplification is detected at 520 nm (FAM™ channel). False negative results due to PCR inhibitors or improper DNA extraction can be ruled out by using the provided internal amplification control, which is detected at 560 nm (HEX™ channel).
TaqMan® based qPCR Assay with FAM™ and HEX™ labeled probes
Applicable in research and industry for the direct detection of Mollicutes (Mycoplasma, Acholeplasma, Spiroplasma) contaminations in cell cultures and biopharmaceuticals. For testing according to EP 2.6.7, USP <1071> and JP G3.
Notice! Not applicable for the ABI 7500/7500 Fast in combination with the AccuSEQ software!
Lyophilized Mix: Primer sets / Nucleotides/ Probes / Polymerase in aliquots of 25 reactions each
Rehydration Buffer
Lyophilized Internal Control DNA
Lyophilized Positive Control
PCR Grade Water
Cycler-based, real-time PCR
PCR reaction tubes
Optional for process validation and EP 2.6.7 / JP G3 compliant testing:
Internal Control DNA Extra (4 vials for 300 µl each of internal amplification control; Cat. No. 11-9905)
10CFU™ Sensitivity Standards available for all EP 2.6.7 / JP G3 listed Mycoplasma species
qPCR cycler with FAM™ and HEX™ filter
Pipetting equipment
Benchtop centrifuge for 1.5 ml reaction tubes
Store the unopened components at +2 °C to +8 °C until the expiry date indicated on the label. Once rehydrated, the components must be stored at ≤ -18 °C.
Yes.
Please note that validation data are provided for information purpose only. EP 2.6.7 clearly states “Where commercial kits are used …, documented validation points already covered by the kit manufacturer can replace validation by the user. Nevertheless, the performance of the kit with respect to its intended use has to be demonstrated by the user (e.g. detection limit, robustness, cross-detection of other classes of bacteria.”. Please feel free to contact us if you need further assistance.
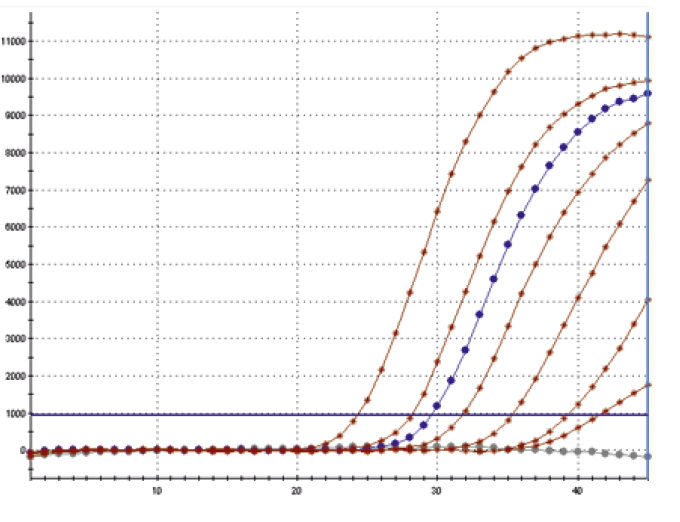
Fig. Amplified dilution series of Mycoplasma fermentans, performed on a
Corbett RotorGene®6000.
Minerva Biolabs offers a free sample of this product for testing:
Request free sample